Patients with persistent, recurrent or metastatic cervical cancer whose tumors express PD-L1 can receive treatment with pembrolizumab plus chemotherapy with or without bevacizumab following approval by the European Commission.

Your AI-Trained Oncology Knowledge Connection!


Patients with persistent, recurrent or metastatic cervical cancer whose tumors express PD-L1 can receive treatment with pembrolizumab plus chemotherapy with or without bevacizumab following approval by the European Commission.

At SGO 2022, Jyoti S. Mayadev, MD presented results of the NRG-GY017 trial of atezolizumab as either an immune primer or with chemoradiation for certain patients with locally advanced cervical cancer.

At SGO 2022, CancerNetwork® spoke with Jyoti Mayadev, MD, about a clinical trial involving immune priming with the PD-L1 inhibitor atezolizumab for patients with locally advanced cervical cancer.
Patients with locally advanced cervical cancer did not see further benefit from the addition of concurrent durvalumab to chemoradiotherapy.
Results of the phase 1 NRG-GY017 trial show promise of atezolizumab as an immune primer in locally advanced cervical cancer.

Updated results from the KEYNOTE-826 study show a favorable risk-benefit ratio for pembrolizumab plus chemotherapy for patients with persistent, recurrent, or metastatic cervical cancer.

An anti-TIGIT/anti–PD-1 combination approach with ociperlimab plus tislelizumab will be examined in a phase 2 study of patients with previously treated recurrent or metastatic cervical cancer.

Patients with recurrent cervical cancer appeared to achieve a promising survival benefit following treatment with cemiplimab.

Pembrolizumab for patients with persistent, recurrent, or metastatic cervical cancer who received chemotherapy with or without bevacizumab had longer progression-free and overall survival vs placebo-treated patients.

In recognition of World Cancer Day, CancerNetwork® spotlights NCCN efforts to reduce disparities in cervical cancer.

In a regulatory update on cemiplimab for advanced cervical cancer, Regeneron Pharmaceuticals announced that the agent’s biologics license application has been withdrawn.

Findings from a phase 2 study demonstrated encouraging clinical activity and a manageable safety profile when patients with advanced cervical cancer were treated with second-line balstilimab and zalifrelimab.

Cervical cancer incidence rates were significantly higher in the lowest-socioeconomic status neighborhoods vs the highest-socioeconomic status neighborhoods in New York City.

Patients with melanoma, head and neck squamous cell carcinoma, and cervical cancer who had not previously received immunotherapy and were treated with lifileucel plus pembrolizumab experienced promising overall response rates compared favorably with historical data on pembrolizumab monotherapy.
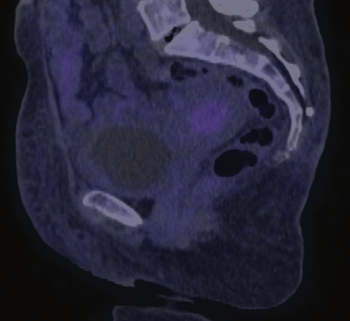
In this edition of Clinical Quandaries Eder A. Arango Bravo, MD, and colleagues present a 63 year old woman who has cervical cancer with kidney failure and additional comorbidities.

Those immunized between the ages of 12 and 13 in England experienced a significant estimated reduction in cervical cancer and grade 3 cervical intraepithelial neoplasia incidence rates compared with unvaccinated women.
Patients with recurrent or metastatic cervical cancer treated with cemiplimab experienced an improved overall survival, progression-free survival, and overall response rate, leading to priority review from the FDA.

Tisotumab vedotin may now be used to treat patients with recurrent or metastatic cervical cancer after the FDA's decision to grant the agent an accelerated approval.

Results of a single-arm study indicate that patients with previously treated metastatic cervical cancer may derive benefit from treatment with balstilimab plus zalifrelimab.

Examination of tisotumab vedotin plus key standard-of-care therapeutics shows promise for the treatment of cervical cancer in the frontline setting and beyond.

Patients with cervical cancer receiving pembrolizumab plus chemotherapy with or without bevacizumab in the frontline setting had improvement in overall survival, potentially representing a new standard of care regimen.

The World Health Organization has published new recommendations to aid in the global prevention of cervical cancer through more accessible screenings and treatments.

Merck’s press release announced positive data from the KEYNOTE-826 trial investigating pembrolizumab plus chemotherapy with or without bevacizumab for patients with cervical cancer.
The anti–PD-1 antibody is being assessed as therapy for patients with recurrent or metastatic cervical cancer after positive results indicate improved responses with the monotherapy.

Awaited outcomes of the phase 3 OUTBACK trial presented at the 2021 ASCO Annual Meeting do not indicate benefit of adjuvant chemotherapy for patients with cervical cancer.